The global stem cell therapy market is experiencing rapid growth. According to authorities like the Korea Health Industry Development Institute, the market is projected to expand from approximately 13.78 billion USD in 2019 to around 23.96 billion USD by 2025.
While domestic companies in South Korea are eager to enter this field, progress has been slow. In contrast, medically advanced nations are making significant strides. The U.S. is accelerating the commercialization of cell and gene therapies through regulatory easing and expedited review processes. Japan has taken an even bolder step by implementing a specialized legal framework for regenerative medicine, creating one of the world’s most efficient environments for clinical trials and commercialization.
Industry reports from Thursday indicate that the U.S. Food and Drug Administration (FDA) introduced the Regenerative Medicine Advanced Therapy (RMAT) designation through the 21st Century Cures Act in 2016.
RMAT streamlines the development and approval processes for regenerative medicine products showing promise in treating serious or rare diseases. This designation covers cell therapies, tissue-engineered products, and gene therapies. It allows for early FDA consultation and offers fast-track benefits such as priority review and accelerated approval.
The U.S. approach balances regulatory flexibility with stringent safety standards. While taking a firm stance against unapproved stem cell procedures, it expedites approvals for treatments backed by solid scientific evidence.
Several therapies in the U.S., including CAR-T, gene therapies, and tissue regeneration treatments, have reached commercialization after receiving RMAT designation. By last year, the FDA had approved RMAT for therapies targeting cancer, rare diseases, skin regeneration, and vascular regeneration.

Experts point to the synergistic collaboration between the National Institutes of Health (NIH), university hospitals, aggressive investments from major pharmaceutical companies, and venture capital funding as key drivers accelerating commercialization.
Companies at the forefront of this race include Mesoblast, bluebird bio, Vertex, and Iovance, all vying for global market dominance with their stem cell, cell therapy, and gene editing technologies.
Japan Pioneers World’s First Regenerative Medicine Special Law
Japan has positioned regenerative medicine as a key future industry to address its aging population and rising healthcare costs. In 2014, the country implemented the Regenerative Medicine Safety Assurance Act and revised the Pharmaceutical Affairs Law. These changes established a conditional and time-limited approval system for regenerative medicine products, allowing early commercialization with post-market data collection requirements once initial safety and efficacy are confirmed.
The nation has strategically fostered stem cell and induced pluripotent stem cell (iPSC) research at a national level. The groundbreaking iPSC technology developed by Professor Shinya Yamanaka of Kyoto University marked a pivotal moment for Japan’s regenerative medicine industry. Yamanaka’s work on reprogramming adult cells to an embryonic stem cell-like state earned him the 2012 Nobel Prize in Physiology or Medicine.
Currently, Japan is aggressively pursuing iPSC-based research for treating retinal diseases, Parkinson’s disease, heart conditions, and spinal cord injuries.
The Japanese approach involves close collaboration between the government, universities, hospitals, and private companies to build a robust clinical network for commercialization. Key institutions leading this effort include the Kyoto University iPSC Research Institute (CiRA), RIKEN, and Keio University.
China has classified stem cell research as a future strategic industry, alongside semiconductors and artificial intelligence (AI). The government is providing substantial funding for large-scale research projects, with stem cell research hubs and bio-clusters emerging in regions like Beijing, Shanghai, and Guangdong.
The scale of stem cell-related clinical trials in China has grown significantly. While some concerns about data reliability persist, the sheer number of clinical studies now ranks second only to the U.S.
South Korea: Strong in Basic Research, but Facing Hurdles
South Korea’s stem cell research capabilities are widely recognized as world-class. Leading companies in stem cell therapy development include Medipost, Cha Biotech, and Kangstem Biotech. Notably, the world’s first stem cell therapy to receive product approval from the Ministry of Food and Drug Safety in 2011 was Hearticellgram-AM, developed by Pharmicell.
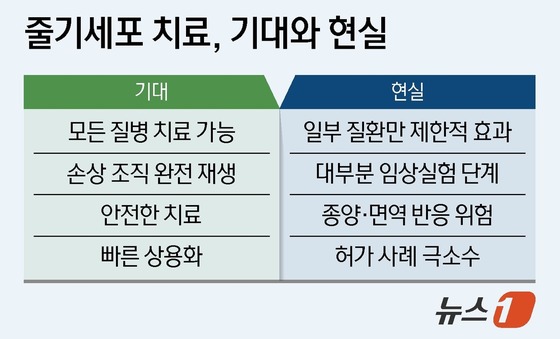
However, the country faces several challenges in advancing stem cell therapies. These include high failure rates in clinical transitions (especially in phases 2 and 3), lack of standardization in safety and efficacy measures, significant cost burdens, and reliance on certain foundational technologies developed elsewhere.
Recent amendments to the Act on Advanced Regenerative Medicine and Advanced Biopharmaceuticals have established a regulatory framework. However, industry insiders still perceive significant regulatory hurdles in clinical and commercialization processes. Additional obstacles include issues surrounding health insurance coverage and concerns about low investment recovery potential.
Despite these challenges, the potential of stem cells in treating intractable diseases, addressing aging, and repairing tissue damage remains immense. Experts believe that if South Korea can enhance its clinical and manufacturing infrastructure, significant opportunities await. Some companies are already making strides in cell culture, contract development and manufacturing (CDMO), and foundational technology fields.
An industry insider noted that the stem cell sector is evolving beyond simple biopharmaceuticals into a platform industry integrating manufacturing, hospitals, data, and AI. For South Korea to remain competitive globally, it must refine its regulatory and insurance systems while fostering a long-term investment ecosystem.