Stem cell therapy has been generating excitement for 20 years, but clinical successes remain few and far between. With the global market projected to reach 27 trillion KRW (about 18.2 billion USD) by 2037, Korean companies are eagerly jumping into the field. However, clinical applications and commercialization are lagging behind technological advances, leaving a persistent gap between expectations and reality.
Stem Cell Therapy: Concept Established, Clinical Applications Limited
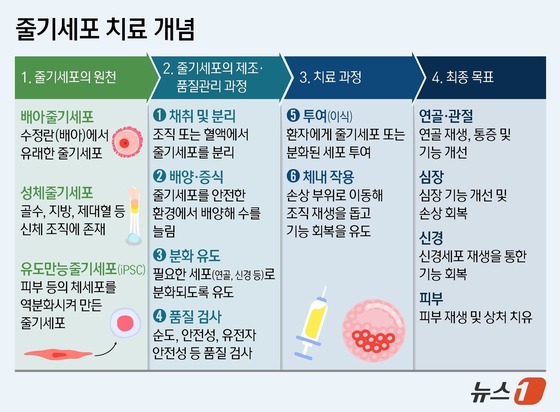
Stem cells can differentiate into specific cell types to repair damaged tissues or organs. Stem cell therapy involves administering these cells or stimulating the body’s own stem cells to treat diseases or restore function.
Different types of stem cells have distinct characteristics and limitations. Embryonic stem cells are highly versatile and genomically stable but ethically controversial. Adult stem cells are safer but have limited applications. Induced pluripotent stem cells (iPSCs) avoid ethical issues but may have genetic instability from reprogramming.
Embryonic stem cells and iPSCs are pluripotent, offering greater therapeutic potential than adult stem cells, but also carry tumor risks.
Currently, established stem cell therapies are rare. A notable example is an U.S. Food and Drug Administration (FDA)-approved mesenchymal stem cell treatment for steroid-resistant graft-versus-host disease. Some therapies, like those for knee cartilage disorders, are already commercialized. Most potential applications remain in clinical trials.
Experts: Limited Concrete Results After 20 Years of Research, Very Few Approvals
Experts are soberly assessing the current state. Dr. Oh Il-hwan of Catholic University notes that despite 20 years of active research, stem cell therapies with proven efficacy remain scarce, with essentially only one approval among countless clinical trials.
He emphasizes that cell therapies aren’t the only solution, pointing to effective new RNA and antibody drugs.
While cell and gene therapy development has surged globally, approval rates remain low. The FDA has become increasingly cautious about single-arm trial approvals, scrutinizing clinical design and data sufficiency more rigorously.
Parkinson’s Trial Shows Functional Recovery: Disease-Specific Approaches Show Promise
Despite prevailing caution, Korea is seeing notable progress. Dr. Kim Dong-wook of Yonsei University recently published results in Cell from a Phase 1/2a trial transplanting embryonic stem cell-derived dopamine precursor cells into Parkinson’s patients.
The year-long study of 12 patients – six receiving low doses (3.15 million cells) and six high doses (6.3 million cells) – showed significant improvements. Long-term Parkinson’s patients regained abilities like playing badminton, table tennis, and even conducting orchestras.
Dr. Kim explains that Parkinson’s unique nature – stemming from selective dopamine cell death – makes it potentially treatable by effectively producing dopamine cells. He predicts Parkinson’s therapies will be commercialized soon, following knee cartilage treatments.
Strong Basic Research Faces Clinical Entry Barriers; Meeting International Standards Crucial
Despite individual successes, high clinical entry barriers hinder broader progress. Dr. Kim Byung-soo of Korea University notes that while Korea excels in basic research, its clinical trial entry rate lags behind competitors.
He warns against dismissing international guidelines like GLP, GCP, and General Motors as excessive regulation, stating this could prevent research from gaining international recognition and harm national competitiveness.
Safety remains a key challenge. The long-term presence of transplanted cells raises concerns about tumor formation, immune reactions, and genetic alterations. Unlike traditional drugs, stem cell therapies require extensive long-term follow-up, but data remains limited.
Unproven Therapies Raise Patient Burden Concerns; Shift to Next-Gen Regenerative Treatments
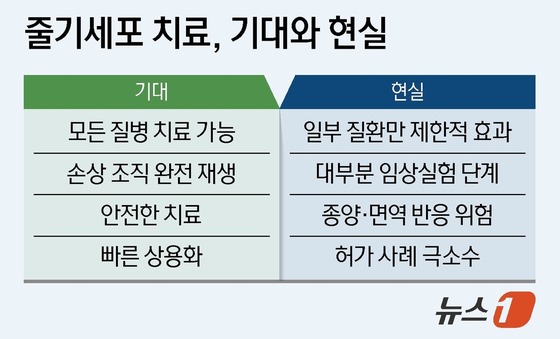
The gap between expectations and reality may burden patients. Despite limited clinical evidence, some pursue costly stem cell treatments. An estimated 30,000 Korean patients travel abroad annually for such therapies, causing 3 trillion to 5 trillion KRW (2.02 billion to 3.37 billion USD) in economic losses.
Experts caution against inflated hopes. Dr. Oh warns that allowing ineffective treatments could violate patients’ health and financial rights. Dr. Kim cautions against viewing stem cell therapy as a cure-all, noting unproven treatments can impose heavy financial burdens.
Yet, the outlook isn’t entirely gloomy. Dr. Oh predicts a shift towards gene-modified cell therapies and molecular regenerative therapies using RNA or peptides to activate endogenous stem cells.
Dr. Kim notes increasing clinical trials of pluripotent stem cells as safety issues are addressed, suggesting commercialization is on the horizon.

