Researchers have uncovered a novel genetic mechanism linked to the development of autism spectrum disorder.
A team led by Dr. Yoo Hee-jung from the Department of Psychiatry at Bundang Seoul National University Hospital revealed on Monday that specific combinations of two genetic mutations can significantly increase the risk of autism, even when individual mutations have minimal impact.
Autism is a complex neurodevelopmental disorder characterized by challenges in social interaction and restricted, repetitive behaviors. Genetic factors are widely recognized as playing a crucial role in its onset.
Individuals with autism often struggle with social communication and exhibit repetitive patterns of behavior. While there’s no known cure, early diagnosis and intervention are critical for managing symptoms and supporting development.
Until now, autism research has primarily focused on single genetic mutations, limiting our understanding of how multiple genes might interact to influence the condition.
To address this gap, Dr. Yoo’s team introduced a groundbreaking gene pair mutation analysis. This innovative approach explores how two genes mutating simultaneously could amplify their association with autism, even if each mutation alone has little effect.
The study analyzed genomic data from 59,168 individuals of diverse ethnic backgrounds, including East Asian families affected by autism and those of European descent. The results were striking: researchers identified six significant gene pairs in East Asians and 156 in Europeans associated with autism.
Intriguingly, these gene pairs were consistently involved in cytoskeletal formation. The cytoskeleton is crucial for maintaining nerve cell structure and facilitating connections between cells. The research team suggests that disruptions to this cellular scaffolding could play a key role in autism’s development.
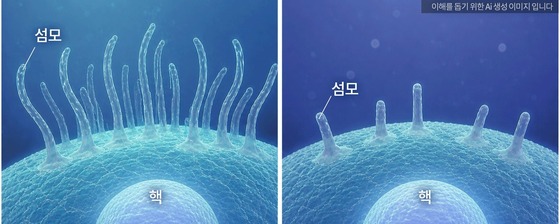
In a collaborative effort with Dr. Sun Woong from Korea University College of Medicine, the team conducted cell experiments that yielded fascinating results. When only one gene of a pair was inhibited, no changes were observed. However, simultaneous inhibition of both genes led to a marked decrease in the formation of cell surface cilia – structures vital for cells to detect external signals and essential for normal brain development.
These findings, the researchers emphasize, demonstrate how the interplay between two genetic mutations can amplify their impact at the cellular level, rather than simply adding their individual effects.
The study also revealed intriguing gender differences. Male autism patients with these specific gene pair mutations exhibited more severe symptoms, while female patients with the same mutations showed no significant differences.
Dr. Yoo highlighted the implications of this research, stating that the findings underscore the need for personalized approaches in autism diagnosis and treatment that account for individual genetic profiles. She added that this work could serve as a cornerstone for developing tailored diagnostic strategies and predictive models for autism.
Dr. Ahn Jun-yong, a co-author of the study, noted that this novel analytical framework could potentially unlock genetic insights into other neurodevelopmental disorders.
The groundbreaking research has been published in the prestigious international journal Genome Biology (Impact Factor 9.4).



