
JW Pharmaceutical is aggressively pursuing the development of innovative hair loss treatments through increased research and development (R&D) investments. The company aims to bolster its competitiveness beyond its current portfolio, which primarily focuses on cholesterol-lowering medications.
According to industry insiders on Tuesday, JW Pharmaceutical’s cholesterol-lowering drug Livalo (pitavastatin) became a blockbuster after its domestic sales launch in 2005, reaching annual sales of 300 billion KRW (approximately 199 million USD) by 2011.
Livalo is renowned for its minimal drug interaction potential, thanks to its metabolism via the UDP-glucuronosyltransferase (UGT) pathway.
This characteristic makes Livalo highly valued in South Korea, where many patients require simultaneous management of multiple chronic conditions such as hypertension and diabetes.
To cater to diverse patient needs, the company has expanded its product lineup.
JW Pharmaceutical has built a comprehensive Livalo family, including Livalozet for patients requiring significant LDL-C reduction, Livalo-V (pitavastatin + valsartan) for those with hypertension and dyslipidemia, and Livalo Hi (pitavastatin + amlodipine + valsartan), a triple combination drug.
Earlier this year, the company launched Livalo Feno, further expanding its range of patient-specific treatment options.
Beyond cholesterol medications, JW Pharmaceutical boasts a diverse portfolio including Hemlibra for hemophilia, Thrupas for benign prostatic hyperplasia, and Actemra for rheumatoid arthritis. The company has also demonstrated market leadership in intravenous fluids with its third-generation three-chamber nutritional solution, Winnerf.
Recognized for its promising pipeline, JW Pharmaceutical is ramping up investments to secure future growth drivers.
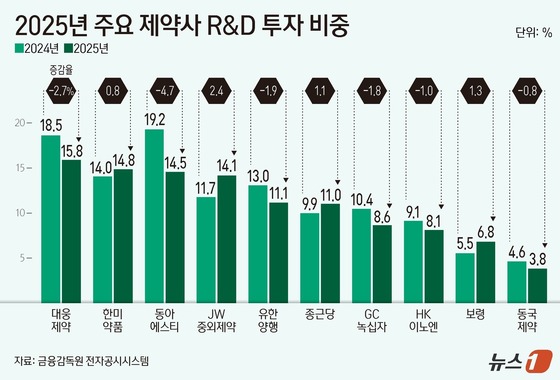
Last year, the company’s R&D investment reached 14.10% of its total revenue, marking a 2.40 percentage point increase from 2021 – the largest jump among domestic pharmaceutical firms.
Hair Loss Treatment Candidate Receives Phase 1 Clinical Trial Approval
This investment boost goes beyond merely increasing clinical trial budgets; it’s intrinsically linked to the company’s long-term growth strategy.
JW Pharmaceutical is leveraging cash flow from its stable existing businesses, such as hospital intravenous fluids, to aggressively develop new drug pipelines for conditions including gout and eye diseases.
Notably, the company’s approach to hair loss treatment, which Targets Wnt signaling pathways, is considered groundbreaking in the domestic pharmaceutical industry.
Recently, JW Pharmaceutical received approval from the Ministry of Food and Drug Safety for the Investigational New Drug (IND) application of its hair loss treatment candidate, JW0061. The company will commence a study involving 104 healthy Korean and Caucasian adults at Seoul National University Hospital.
The clinical trial aims to evaluate JW0061’s safety and tolerability through topical application to the scalp, while also assessing its pharmacokinetic properties, including absorption and metabolism in the body.
JW Pharmaceutical has secured patent registrations for JW0061 in several countries, including South Korea, Japan, China, Australia, and Brazil. Last month, the company completed U.S. patent registration, ensuring exclusive rights to its proprietary technology in the American market until 2039.
Based on the Phase 1 trial results, JW Pharmaceutical plans to refine its subsequent clinical development strategy and systematically review indications and development plans.
A JW Pharmaceutical spokesperson stated that the IND approval marks a significant milestone as JW0061 transitions from preclinical to clinical stages. It is committed to advancing the clinical development without delays to establish JW0061 as a global innovative hair loss treatment.
