
A groundbreaking study by South Korean researchers has revealed that long before lung cancer becomes visible as a tumor, mutated cells engage in a complex dialogue with surrounding cells, creating an environment conducive to cancer growth. This discovery marks a significant shift from traditional post-diagnosis treatments, potentially paving the way for early intervention strategies.
On Wednesday, the Gwangju Institute of Science and Technology (GIST) announced that Professor Choi Jin-wook’s research team, in collaboration with Professor Lee Joo-hyun’s team from Memorial Sloan Kettering Cancer Center (MSK) in the U.S., has mapped out the cascade of cellular interactions during the early stages of lung cancer development.
The findings were published online in the prestigious scientific journal Nature on April 22.
Lung adenocarcinoma (LUAD), a highly lethal form of cancer, often goes undetected in its early stages due to a lack of symptoms. Consequently, most patients are diagnosed when the disease has already progressed significantly, limiting treatment options.
While previous research has focused on how genetic mutations in lung stem cells (AT2) lead to cancer, the specific process by which mutated cells transform surrounding healthy tissue into a cancer-friendly fibrotic niche remained elusive.
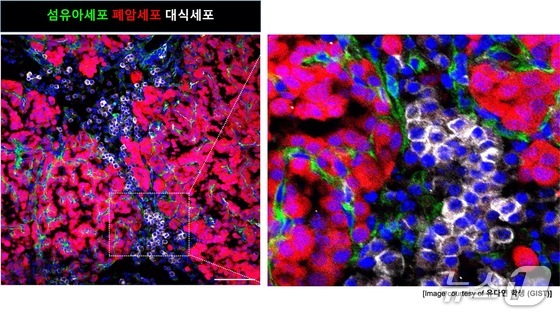
Using mouse models and innovative three-dimensional (3D) lung organoids, the research team tracked the intricate cellular communications occurring at the onset of cancer development.
Their findings revealed a three-stage process in lung cancer development. Initially, mutated lung stem cells flood their environment with a signaling molecule called amphiregulin (AREG), triggering aggressive responses in neighboring cells.
In the second stage, surrounding fibroblasts, responding to these signals, lose their tissue-repairing abilities and transition into a fibrotic state, effectively hardening the tissue. This transformation is analogous to converting fertile soil into a specialized environment optimized for cancer cell growth.
The final stage sees this fibrotic environment attracting immune cells (macrophages), which amplify the inflammatory response. This inflammation, in turn, accelerates the malignant transformation of mutated cells, creating a self-perpetuating cycle. This vicious feedback loop between cancer cells and their environment ultimately leads to full-blown tumor development.
Crucially, the team discovered that blocking the amphiregulin signaling pathway, a key link in this cascade, through genetic and pharmacological interventions significantly inhibited the formation of the fibrotic microenvironment, effectively preventing early lung cancer onset.
To validate these findings in a clinical context, the research team collaborated with Professor Park Moo-seok’s team at Yonsei University Severance Hospital. They developed a 3D organoid lung cancer model that simulates patient conditions, allowing for ex vivo assessment of their discoveries.
Their experiments confirmed that the fibrotic microenvironment is indeed induced within actual lung tissue, even in models with mutations in KRAS, a key gene regulating cell growth and division.
Professor Choi emphasized the significance of this research, stating that this study represents a paradigm shift in cancer treatment. By identifying new therapeutic targets that can prevent cancer at its earliest stages, they’re opening doors to next-generation preventive measures and precision-targeted therapies for lung cancer.
The study was co-led by Professor Choi and Professor Lee from Memorial Sloan Kettering Cancer Center (MSK), with GIST’s Lee Hye-young and MSK doctoral student Erik Cardoso as lead authors.
