
Surgical techniques in the medical field are evolving rapidly. While minimizing scarring on patients’ bodies, doctors are simultaneously broadening and refining their view of lesions.
Endoscopy is at the forefront of this trend. In orthopedics, it has transitioned from a supplementary tool to a key instrument that’s revolutionizing treatment paradigms. In this landscape, Meditech is spearheading the development of domestically produced endoscopic orthopedic surgical devices.
According to industry sources, including the Medical Device Research and Development Project Group, Meditech is a medical device startup founded in 2020 that leverages technology from the Korea Electric Research Institute (KERI).
Founder and CEO Lee Chi Won, a Seoul National University graduate in mechanical and aerospace engineering, earned his Ph.D. in bioengineering from the same institution. Before developing endoscopes at KERI, he served as a senior researcher at Seoul National University’s Medical Research Institute.
With Japanese firms like Olympus and Fujifilm controlling about 90% of the global gastrointestinal endoscope market, Lee has stepped up to the challenge. Meditech’s growth centers on developing a smart, flexible endoscope that integrates electric operation with AI lesion detection.
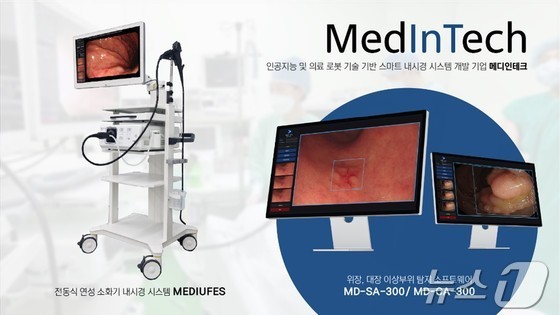
Conventional endoscopes are primarily mechanical, with numerous components in the control unit that doctors manually manipulate to guide the endoscope through a patient’s body.
Extended use of these devices can be physically taxing for physicians. Meditech has addressed this by redistributing core components throughout the main body connected to the endoscope, effectively halving the control unit’s weight.
During examinations, if abnormalities are detected, doctors can customize the thickness, color, and shape of the on-screen alert box. They can also adjust detection sensitivity for both stomach and colon examinations. Additional customizable features include alarm types, volume, and resolution settings, allowing for an optimized endoscopic procedure environment.
An industry expert noted that Meditech’s AI software, developed and validated in partnership with gastroenterology teams at top-tier university hospitals, could significantly enhance the reliability of endoscopic examinations.
Meditech envisions a future where AI and robotics technologies transcend current limitations in endoscopic medicine, setting new global standards for endoscopic diagnosis and treatment.
Over the past two years, Meditech has conducted approximately 500 clinical trials in collaboration with six major South Korean university hospitals, including Seoul National University Hospital, validating its technological prowess.
The company has also been successful in attracting investments. On May 13, 2024, it secured a Series B investment of about $15 million, bringing its total funding, including Series A (approximately $6 million) and seed investments, to over about $21 million.
Notably, Meditech is engaged in several national projects, including a multi-agency medical device research and development initiative valued at about $7.125 million. The company is accelerating the development of its innovative endoscopic system by advancing electrification modules and AI-based technologies.
Meditech is currently navigating domestic and international regulatory approval processes. The company is also in discussions to implement its products in clinical settings across Southeast Asia, South America, and the United States.
A Meditech spokesperson emphasized that their goal is to provide an integrated solution encompassing all aspects of endoscopic procedures. The company aims to evolve into a comprehensive platform supporting the entire process from diagnosis to treatment.
