A groundbreaking study has shown that combining targeted therapies for HER2-positive gastric cancer significantly boosts treatment effectiveness.
On Thursday, researchers led by Dr. Lee Geun-wook from the Hematology and Oncology Department at Bundang Seoul National University Hospital announced that administering trastuzumab deruxtecan (Enhertu) together with pertuzumab (Perjeta) to mice with HER2-positive tumors resulted in an impressive 81% reduction in tumor size compared to untreated controls.
HER2-positive gastric cancer is caused by an overproduction of the HER2 protein, which accelerates cell growth and division. This aggressive form of cancer affects approximately 20% of gastric cancer patients and typically has a grim prognosis, with average survival rates of only 16 to 20 months.
The standard treatment for HER2-positive gastric cancer involves targeted therapies, with trastuzumab deruxtecan being a key player.
Unlike traditional chemotherapy, which can harm healthy cells, targeted therapies precisely attack specific molecules or proteins on cancer cells. This approach generally offers better efficacy and more manageable side effects.
Trastuzumab deruxtecan works by delivering chemotherapy agents directly to HER2 proteins on cancer cell surfaces.
However, some patients have shown diminished response to targeted therapies, and others develop resistance during treatment. This occurs because HER2 cancer cells team up with another protein, HER3, to enhance their survival mechanisms.
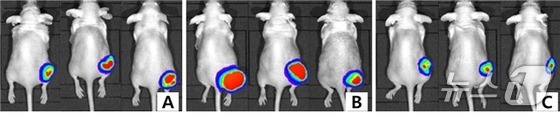
To explore the potential of combining trastuzumab deruxtecan and pertuzumab, the research team implanted HER2-positive tumors in 12 experimental mice.
The mice were then divided into three groups of four: one receiving trastuzumab deruxtecan alone, another receiving pertuzumab alone, and a control group receiving no treatment. The researchers administered the drugs weekly and monitored tumor sizes over a six-week period.
The results were striking: the combination therapy group showed an average tumor size of 110.4 mm³, a remarkable 81% reduction compared to the untreated control group (580.4 mm³).
In comparison, the trastuzumab deruxtecan monotherapy group had tumors measuring 263.2 mm³ (a 54.7% reduction), while the pertuzumab monotherapy group showed tumor sizes of 446.9 mm³ (a 23% reduction).
Weekly measurements revealed a consistent decrease in tumor size for the combination therapy group, while tumors in the other groups continued to grow.
Dr. Kang Min-soo explained the mechanism behind the combination therapy: Pertuzumab blocks the interaction between HER2 and HER3, allowing trastuzumab deruxtecan to more effectively penetrate cancer cells and trigger cell death. The two drugs work synergistically, amplifying the overall treatment efficacy.
Dr. Lee emphasized the significance of these findings: Targeted therapies have recently faced challenges due to resistance, leading to reduced effectiveness. This combination approach represents a major breakthrough, opening up new possibilities for maximizing treatment outcomes in HER2-positive gastric cancer patients.
The study has been published in Molecular Cancer Therapeutics, a prestigious journal of the American Association for Cancer Research (AACR) with an impact factor of 5.1.
