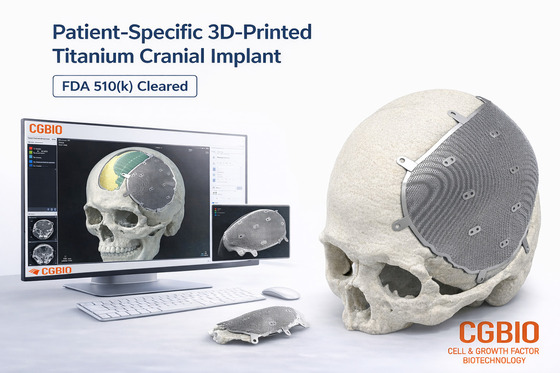
CG Bio announced on Wednesday that its patient-customized titanium implant, EasyMade TI, received 510(k) approval from the U.S. Food and Drug Administration (FDA) on April 9.
The FDA 510(k) is a key pre-market approval process that demonstrates a new medical device’s equivalence to existing approved products for sale in the U.S. This marks the first time a South Korean company has obtained FDA 510(k) approval for a patient-customized titanium implant targeting the U.S. market.
EasyMade TI is a patient-customized titanium implant designed using the patient’s computed tomography (CT) data. It was developed to reconstruct skull and non-load-bearing craniofacial defects. The implant is made from medical-grade titanium alloy using laser powder bed fusion (LPBF), an advanced metal three-dimensional (3D) printing technology.
The product’s supply process is also considered a competitive advantage. When U.S. medical professionals send a patient’s CT images, CG Bio’s design center in South Korea creates a customized design and delivers the product to U.S. hospitals within five days. After in-hospital sterilization, the implant is ready for immediate surgical use.
This approval is expected to be a game-changer for CG Bio’s expansion into the U.S. customized implant market. The demand for reconstruction following trauma, tumor resection surgery, and decompressive craniectomy is steadily growing in the U.S., along with increasing interest in customized reconstruction solutions tailored to patients’ unique anatomical structures.
Yu Hyun-seung, Chief Executive Officer (CEO) of CG Bio, stated that the FDA 510(k) clearance for EasyMade TI is a significant milestone that demonstrates SG Bio’s global competitiveness in patient-customized reconstruction solutions. It plans to aggressively expand our customized skull and craniofacial reconstruction implant business in the U.S. market and actively pursue global approvals and commercialization for the future product lines.
