New research reveals why cold sores appear around the lips when the herpes virus activates: our body’s innate immune sensors recognize repetitive sequences in the virus’s DNA.
A team led by Professor Lee Sang-jun from Ulsan National Institute of Science and Technology (UNIST) announced their findings on Monday. In collaboration with researchers from Sungkyunkwan University, Jeju National University, and the IBS Korean Virus Research Institute, they identified that the poly(T) repetitive sequence on herpes virus DNA triggers an immune response.
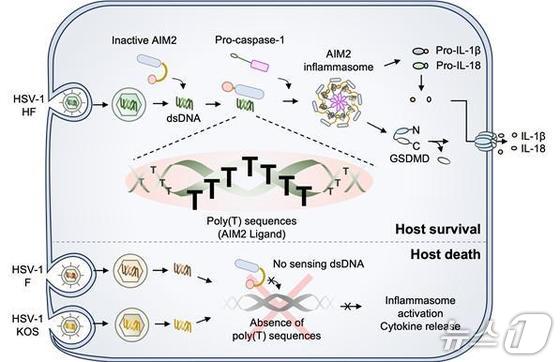
Herpes simplex virus type 1 hides in nerve ganglia, evading immune system attacks. It then exploits weakened immune surveillance to infect host skin cells. During this process, the protein AIM2 acts as a sensor, detecting the virus within host macrophages.
The study shows that AIM2 recognizes long repetitive segments of thymine (T) nucleotides in viral DNA to detect the virus.
Even within the same herpes simplex virus type 1 strain, AIM2 only activates when the viral DNA contains the poly(T) sequence, triggering inflammation and infected cell death.

When this sequence was introduced into other strains, it induced a new immune response. The researchers also confirmed a length dependency, where longer repetitive sequences elicited stronger reactions.
Animal experiments revealed that infection with viruses containing the poly(T) repetitive sequence led to inflammation and suppressed viral replication. Conversely, viruses lacking this sequence caused minimal immune response and rapid viral growth.
The team also discovered that this poly(T) repetitive sequence is widely conserved among various infectious disease viruses, including coronaviruses.
Professor Lee Sang-jun noted that therapies that modulate immune sensor activity, rather than directly attacking viruses, are emerging as promising alternatives. This discovery lays the groundwork for developing tailored immune-modulating drugs.
The findings were published online on April 13 in the prestigious journal Nature Communications.
