A groundbreaking subcutaneous influenza vaccine for dogs is making waves in the veterinary field.
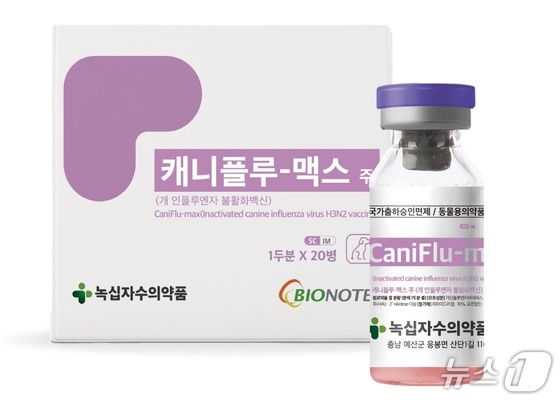
Green Cross Veterinary Products (GCVP), led by CEO Na Seung Sik, announced on Tuesday that it has secured additional approval from South Korea’s Animal and Plant Quarantine Agency for its inactivated canine influenza vaccine, CaniFlu-max.
This new approval expands administration options beyond intramuscular injections to include subcutaneous injection, giving veterinarians greater flexibility in tailoring treatments to each dog’s needs and owner preferences.
This development is particularly beneficial for dogs that are difficult to restrain or for owners who are uncomfortable with intramuscular injections.
CaniFlu-max has been in use since its initial approval in 2009, with a 17-year track record of clinical data.
With more than 200,000 dogs vaccinated annually, the vaccine’s safety profile is notable: only one adverse event has been reported per 200,000 doses administered, earning it the World Health Organization’s “Very Rare” safety classification.
The vaccine’s production method also sets it apart. CaniFlu-max uses cell culture technology, eliminating the risk of viral mutations associated with egg-based cultivation. Studies suggest that these results stem from an antigen structure more closely aligned with circulating viruses, potentially leading to stronger neutralizing antibody responses.
Canine influenza presents symptoms similar to human flu, and veterinarians in South Korea generally recommend annual vaccination. For multi-dog households or environments with higher exposure risk, vaccination against canine influenza is considered essential.
A spokesperson for GCVP said the approval is intended to enhance the clinical utility of an already-established vaccine, adding that the company remains focused on developing flexible prevention and treatment options to address the diverse needs of veterinary practices nationwide.