Onconic Therapeutics recently presented the results of a large-scale Real-World clinical study on their potassium-competitive acid blocker (P-CAB) Zacubo at the 2026 Digestive Disease Week (DDW 2026) in Chicago.
The study aimed to validate Zacubo’s efficacy and safety across diverse patient groups in real clinical settings and explore its potential to address unmet needs in patients unresponsive to existing treatments.
At DDW 2024 and 2025, Onconic Therapeutics unveiled Phase 3 results for erosive gastroesophageal reflux disease and gastric ulcers, respectively. This year’s presentation of extensive Real-World Data further bolsters Zacubo’s clinical credibility.
The multi-center study, conducted from 2024 to 2025, involved about 300 private clinics across South Korea. It tracked over 5,500 gastroesophageal reflux disease patients prescribed Zacubo 20 mg for four weeks, analyzing real-world treatment outcomes.
The study’s significance lies in its corroboration of previous clinical trial results within general practice settings, involving multiple centers and a substantial patient cohort.
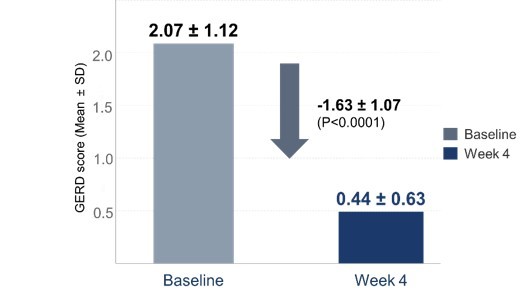
Analysis of the GERD questionnaire, which assesses typical symptoms like heartburn and acid reflux on a 0-5 scale, revealed statistically significant improvement. Average scores dropped from 2.07 pre-treatment to 0.44 after four weeks (P < 0.0001).
Notably, patients who had not responded to existing proton pump inhibitors (PPIs) or acid suppressants showed marked RDQ score reductions. This Real-World data suggests Zacubo’s potential to address symptoms resistant to current treatments, fulfilling an unmet medical need.
Patient satisfaction was remarkably high, with 93.4% reporting being satisfied or very satisfied with the treatment. The adverse drug reaction (ADR) rate was a mere 0.05%, indicating a favorable four-week safety profile.
Patients highlighted Zacubo’s key advantages: the ability to take it regardless of meals and its effectiveness in alleviating previously unresolved symptoms within just four weeks.
This study demonstrates that switching to Zacubo could be an effective strategy for patients with a history of PPI and acid suppressant use, as evidenced by extensive real-world clinical data.
An Onconic Therapeutics spokesperson stated that the real-world efficacy of switching to Zacubo has been validated by large-scale data, offering new options for GERD patients. This empirical evidence has propelled Zacubo to blockbuster status, exceeding 20 billion KRW (about 13.7 million USD) in quarterly prescriptions within just six quarters post-launch. We aim to leverage these findings to expand Zacubo’s impact in both domestic and international markets.”
Nesuparib Shows Promise in Suppressing Pancreatic Cancer Metastasis
In related news, Onconic Therapeutics presented findings from a preclinical study on their next-generation dual-inhibitor cancer drug candidate, Nesuparib, at the 2026 American Association for Cancer Research (AACR) meeting in San Diego.
The conference highlighted Nesuparib’s ability to inhibit cancer metastasis and its anticancer effects beyond traditional PARP inhibitors, demonstrating efficacy independent of BRCA gene mutations.
Pancreatic cancer, known for its aggressive metastasis and limited treatment options, has a dismal five-year survival rate of just over 10%. This rate plummets to 2-3% in cases of distant metastasis, underscoring the urgent global need for novel treatment strategies.
Nesuparib, a first-in-class candidate, is designed to simultaneously inhibit PARP and suppress Tankyrase. Conventional PARP inhibitors have shown limited effectiveness, primarily in patients with DNA repair defects like BRCA mutations. In South Korea, only about 5% of pancreatic cancer patients carry BRCA mutations.
Contrastingly, this study demonstrated Nesuparib’s anticancer and anti-metastatic effects in pancreatic cancer models without BRCA mutations (wild-type), potentially broadening the drug’s applicability to a wider patient population.



